Protein Domains
Introduction
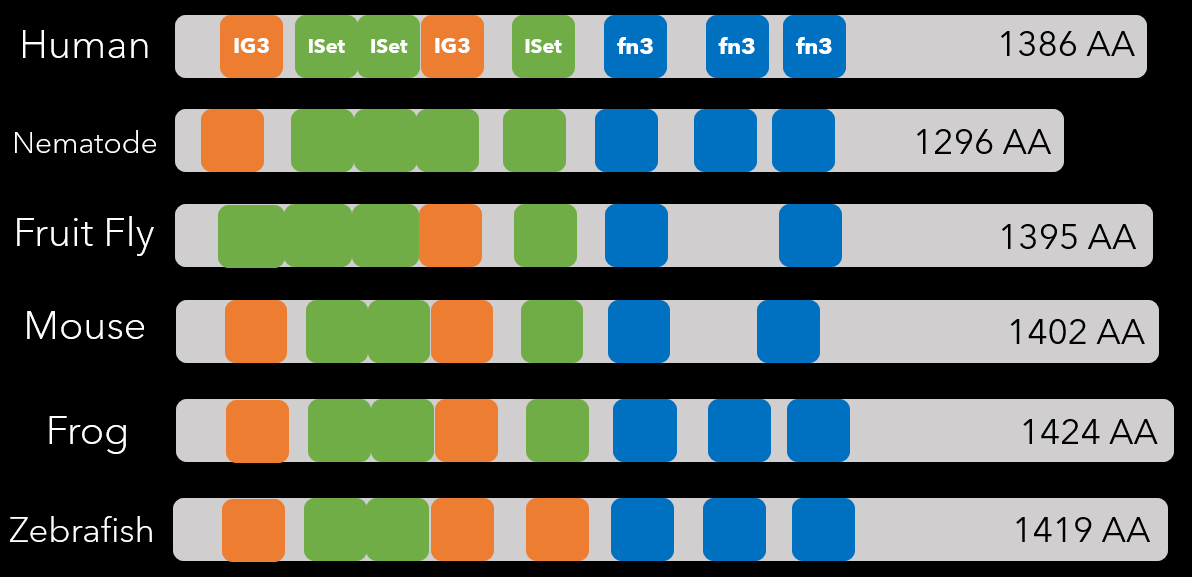
Protein domains are the functional units of a protein sequence. Some complex genes are made up of multiple protein domains that each serve a different purpose in contributing to the overall protein structure and function [6]. Throughout evolutionary time, some domains can be duplicated, deleted, or rearranged by small mutations. Exploring the differences (and similarities) between homolgous protein sequences can provide clues to how the protein products function within different organisms. Conserved protein domains can be identified using bioinformatics tools like PFAM and SMART, as shown below [3].
Results
IG3 - Immunoglobin Domain
ISet - Immunoglobin I-Set Domain
fn3 - Fibronectin Type 3 Domain
ISet - Immunoglobin I-Set Domain
fn3 - Fibronectin Type 3 Domain
Discussion
The IG3 domain is a subgroup of the immunoglobin protein domains. These domains function in cell adhesion molecules, cytokine-cytokine receptors, and immune system response. They are frequently found in sequence with I-set domains [5]. The I-Set domain is also found in many cell-cell adhesion molecules, of which we know the ROBO3 protein is one based on the gene ontology results. Interestingly, the I-set domain is also found in axonin-1, a neuronal adhesion molecule and semaphorin 4D, a signaling molecule involved in axonal guidance [2, 1]. This supports the idea the I-set domain is important for genes found within axons specifically - and is further supported by the fact that the I-Set domain is the most conserved in number throughout the homologs. Finally, the fibronectin type 3 domain is a plasma protein that is also implicated in cell ahesion and cell migration [4]. It also is found to be related to protein binding - another one of ROBO3's GO terms, and predicts possible function in linking ROBO and SLIT proteins (ROBO's ligand).
References
- Esnouf, Robert M. et al. "Structure Determination Of Human Semaphorin 4D As An Example Of The Use Of MAD In Non-Optimal Cases". Acta Crystallographica Section D Biological Crystallography, vol 62, no. 1, 2005, pp. 108-115. International Union Of Crystallography (Iucr), doi:10.1107/s0907444905034992. Accessed 20 Mar 2020.
- Freigang, Jörg et al. "The Crystal Structure Of The Ligand Binding Module Of Axonin-1/TAG-1 Suggests A Zipper Mechanism For Neural Cell Adhesion". Cell, vol 101, no. 4, 2000, pp. 425-433. Elsevier BV, doi:10.1016/s0092-8674(00)80852-1. Accessed 20 Mar 2020.
- Lee, D., Redfern, O. & Orengo, C. Predicting protein function from sequence and structure. Nat Rev Mol Cell Biol 8, 995–1005 (2007). https://doi.org/10.1038/nrm2281
- "SMART: FN3 Domain Annotation". SMART, 2020, http://smart.embl.de/smart/do_annotation.pl?BLAST=DUMMY&DOMAIN=FN3.
- "SMART: IG Domain Annotation". SMART, 2020, http://smart.embl.de/smart/do_annotation.pl?DOMAIN=SM00409.
- "What Are Protein Domains?". EMBL-EBI Train Online, 2020, https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains.